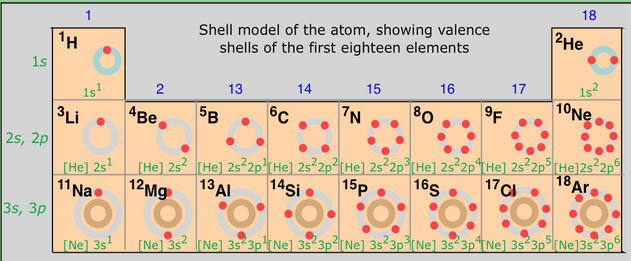
This table is also available with a white background here for better toner/ink conservation. For best printing results, choose Landscape and ‘Fit’ for the size option. Similar electron configurations within a group of the Periodic Table can be emphasised with a simpler representation in terms of the previous noble gas. It is also available for download in PDF format for printing. Click the image for full size and save to your computer. It has a resolution of 1920×1080 and is easily readable on most screens. Searchbuild circle Toolsfact check Homeworkcancel Exit Reader View school Campus Bookshelves menu book Bookshelves perm media Learning Objects login Login how reg Request Instructor Account hub Instructor CommonsSearch Downloads expand more Download Page PDF Download Full Book PDF. The image above is suitable for widescreen desktop wallpaper for your electronic devices. These are denoted with an asterisk on the table and the value listed is a predicted value. This shorthand uses the name of the previous row’s noble gas in brackets to represent the part of the electron configuration is identical to the noble gas’ electron configuration.Įlements greater than 104 are unknown or unverified. A version of this table with the correct atomic masses can be found here.Įach configuration is written in the noble gas shorthand. Therefore the atoms within a certain group grow larger from top to bottom of the periodic table. As the number of period increases a new electron shell is added. EDIT: This table, while looking very nice, contains some minor errors in the values for atomic mass of some elements. The periodic table arranges elements into groups with similar chemical properties and periods with identical number of shells Figure: Periodic table of elements. 6 electrons are in p-orbitals.Color Periodic Table wallpaper with electron configurations and dark background.Similarly, other 2 electrons are in s-orbitals of the 2nd energy shell, and.Here, 2 electrons are in s-orbitals of 1st energy shell.The 2, 2and 6(in red color) are the number of electrons present in that particular orbitals.The electron configurations are written in the noble gas notation. The s and p indicates that the electrons are lying in that orbitals. Printable Periodic Table With Electron Configurations Todd Helmenstine This periodic table contains each element's atomic number, atomic mass, symbol, name, and electron configuration.Elements are placed in order on the periodic table based on their atomic number, how many protons they have. 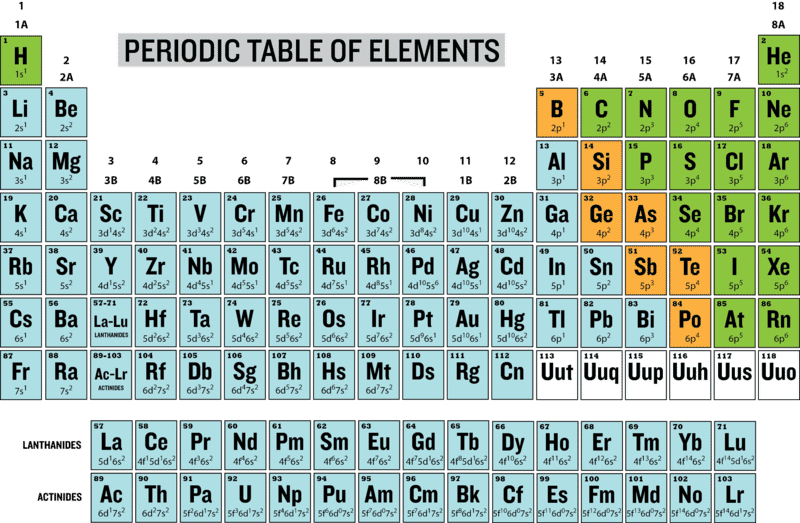 
The number 1, 2 and 2 (in red color) indicates the Principal quantum number. Electron configurations and the periodic table.Through electronic configuration, the chemical properties of elements can be predicted by knowing their valence electrons.ġs 2 2s 2 2p 6 simply indicates the following things.Number of electrons present in s, p, d and f orbitals can be found using electron configuration.Electron configuration is very important to know where the electrons are located around the atoms. Each shell can contain only a fixed number of electrons: the first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on.f-orbitals Why is Electron configuration important?Įlectronic configuration is important in the following ways.These regions around the nucleus are known as orbitals or subshells. A layered arrangement of shells, where a specific number of electrons orbit around the nucleus. For example, here you can see that the blue and yellow color regions around the nucleus show a maximum probability of finding the electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
